Researchers in the U.S. have shown both preventive and treatment effects of an antibody cocktail in animal models of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) – the agent that causes coronavirus disease 2019 (COVID-19). A combination of two monoclonal antibodies significantly reduced airway viral load and virus-induced pathology in models of both mild and severe disease.
*
More information …
REGEN-COV™ (casirivimab and imdevimab) is an investigational medicine used to treat mild to moderate symptoms of COVID-19 in non-hospitalized adults and adolescents (12 years of age and older who weigh at least 88 pounds [40 kg]), and who are at high risk for developing severe COVID-19 symptoms or the need for hospitalization. REGEN-COV is investigational because it is still being studied. There is limited information known about the safety and effectiveness of using REGEN-COV to treat people with COVID-19.
*
EMERGENCY USE AUTHORIZATION (EUA) OF CASIRIVIMAB AND
IMDEVIMAB
https://www.fda.gov/media/143892/download
*
Antibody cocktail
https://www.youtube.com/watch?v=mvfomfrxCGQ
https://www.forbes.com/sites/brucelee/2020/11/22/regeneron-antibody-cocktail-for-covid-19-coronavirus-gets-fda-emergency-use-authorization/?sh=d1c31dccba8c
*
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders.
More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
Casirivimab and imdevimab is a cocktail of two monoclonal antibodies (also known as REGN10933 and REGN10987, respectively) and was designed by Regeneron scientists to block infectivity of SARS-CoV-2, the virus that causes COVID-19. They evaluated thousands of fully-human antibodies produced by the company’s proprietary VelocImmune® mice, which have been genetically modified to have a human immune system, as well as antibodies identified from humans who have recovered from COVID-19.
https://www.roche.com/media/releases/med-cor-2021-03-23.htm
https://www.roche.com/
*
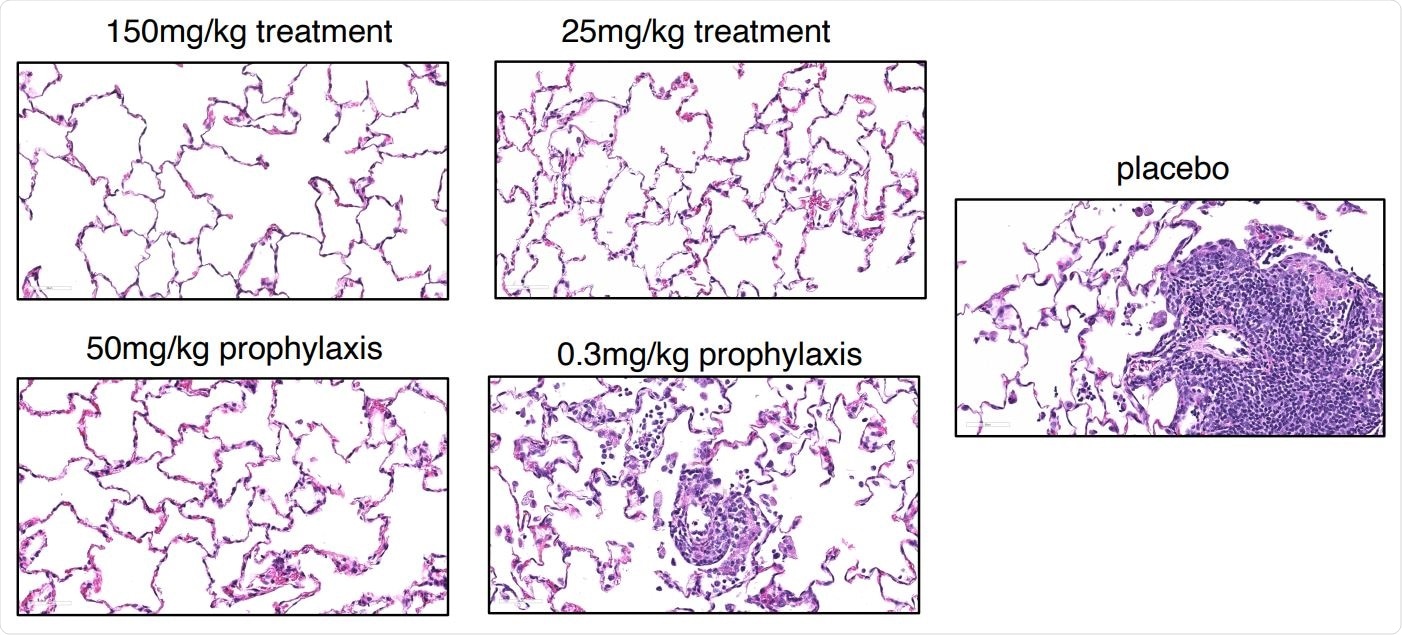
The authors say the findings point to the therapeutic potential of REGN-COV2 in both protecting against SARS-COV-2 infection and treating COVID-19.
They also say the effect the antibody cocktail had on viral RNA levels in NP and BAL swabs suggests not only the potential to prevent disease following exposure but also to limit further transmission. The authors say their findings provide evidence that this REGN-CoV-2 therapy, which is currently being tested in clinical trials of COVID-19, may provide significant clinical benefit in both the prevention and treatment setting.
Compared with placebo-treated animals, viral RNA was almost wholly ablated in most animals, indicating that the therapy can almost completely stop infection becoming established. This effect was seen across all NP and BAL measurements, indicating that viral loads were significantly reduced in both the upper and lower airways. To assess the treatment effect of REGN-CoV-2, the macaques were given 25mg/kg or 150mg/kg doses one day following challenge with 1×10^6 PFU of the virus.
To assess the prophylactic effect of REGN-CoV-2 in the context of more severe disease, the golden hamster was given 50, 5, or 0.5mg/kg of the drug two days prior to challenge with 2.3×10^4 PFU of virus.
- Kyratsous C, et al. REGN-COV2 antibody cocktail prevents and treats SARS-CoV-2 infection in rhesus macaques and hamsters. bioRxiv 2020. doi: https://www.biorxiv.org/content/10.1101/2020.08.02.233320v1
*
On June 3, 2021, the U.S. Food and Drug Administration updated the Emergency Use Authorization for REGEN-COV, lowering the dose to 1,200 mg (600 mg casirivimab and 600 mg imdevimab) is half the dose originally authorized. As part of the updated EUA, REGEN-COV should be administered by intravenous infusion; subcutaneous injections are an alternative when IV infusion is not feasible and would lead to a delay in treatment.
.
On May 26, 2021, the U.S. CDC announced that the combined frequency of the P.1 variant (Brazil) and the B.1.351 variant (South Africa) now exceeds 10% in Arizona, California, Florida, Indiana, Oregon, and Washington. REGEN-COV is an alternative monoclonal antibody therapy currently authorized for the same use as bamlanivimab and etesevimab administered together. Based on similarities in vitro assay data currently available, REGEN-COV is likely to retain activity against the P.1 and B.1.351 variants. The U.S. FDA recommends that health care providers in Arizona, California, Florida, Indiana, Oregon, and Washington use this alternative authorized monoclonal antibody therapy until further notice.
June 16, 2021 – Regeneron Pharmaceuticals, Inc. confirmed positive results from the largest trial assessing any monoclonal antibody treatment in patients hospitalized with severe COVID-19. The UK RECOVERY trial found that adding investigational REGEN-COV™ to usual care reduced the risk of death by 20% in patients who had not mounted a natural antibody response on their own against SARS-CoV-2, compared to usual care on its own.
June 9, 2021 – Canada Health Authorized By Interim Order (249830) Casirivimab and imdevimab for use concerning the COVID-19 pandemic, per section 5 of the Interim Order Respecting the Importation, Sale, and Advertising of Drugs for Use concerning COVID-19.
June 4, 2021 – Regeneron Pharmaceuticals, Inc. announced the U.S. Food and Drug Administration (FDA) updated the Emergency Use Authorization (EUA) for REGEN-COV™, lowering the dose to 1,200 mg (600 mg casirivimab and 600 mg imdevimab), which is half the dose.
This COVID-19 Antibody Cocktail’s development and manufacturing has been funded in part with federal funds from the Biomedical Advanced Research and Development Authority under OT number: HHSO100201700020C.
The concept that drug cocktails can prevent viral escape has previously been demonstrated for traditional antiviral drugs used to treat HIV and other viruses. Regeneron now reports the fundamental realization that this can also be true for antibody-based therapies, as reported in a paper published in Science on August 21, 2020, entitled “Antibody Cocktail to SARS-Cov-2 Spike Protein Prevents Rapid Mutational Escape Seen with Individual Antibodies,” which further defines the protective value of the multiple-antibody approach against SARS-CoV-2 specifically.
Regeneron’s REGEN-COV Antibody Cocktail is a combination of monoclonal antibodies casirivimab and imdevimab, explicitly designed to block the infectivity of SARS-CoV-2, the coronavirus that causes COVID-19. This was the first antibody therapy to demonstrate an anti-viral effect in patients hospitalized with COVID-19.
Regeneron’s core technologies allow for the rapid and efficient generation of these protective anti-viral antibodies outside of the body, derived from genetically humanized mice or convalescent humans. The resulting antibodies correspond to the most potent anti-viral antibodies that could be elicited by a vaccine or through exposure to a pathogen.
These REGEN-COV antibodies can be delivered to people via injection, providing “passive immunity” and immediately protecting the disease. However, they must be re-administered to remain effective over time. These antibodies can also treat an existing infection, unlike vaccines, which can only be used preventatively.
Regeneron scientists evaluated thousands of fully human antibodies produced by the company’s proprietary VelocImmune® mice, which have been genetically modified to have a human immune system, as well as antibodies isolated from humans who have recovered from COVID-19.
https://www.precisionvaccinations.com/vaccines/regen-cov-antibody
*
Related articles
FDA coronavirus patients to be treated with blood from survivors
Dr Dan Lee Dimke PhD – COVID-19 virus
Ivermectin in SA Doctors
Ivermectin – FLCCC
Ivermectin – Covid-19 – Prof Thomas Borody
Ivermectin – India and Brittain
COVID-19: The Ivermectin African Enigma