Why are bats the reservoirs of so many EIDs? Bats have several unique features that may account for their importance in EID transmission and maintenance. Bats are the second largest order of mammals. Currently, there are ∼1,200 recognized bat species worldwide, accounting for approximately 25% of all mammalian species. The diversity of bat species alone, along with their worldwide distribution, contributes to the biodiversity of their pathogens.

*
Emerging Infectious Diseases
*
Bartonella quintana infection (historically called ‘trench fever’) is a vector-borne disease primarily transmitted by the human body louse Pediculus humanus humanus. The infection is associated with a wide variety of clinical conditions, including chronic bacteraemia, endocarditis, lymphadenopathy and bacillary angiomatosis. Since the 1990s, it has been recognised as a reemerging pathogen among impoverished and homeless populations — so-called ‘urban trench fever’ — living in unsanitary conditions and crowded areas predisposing them to infestation with ectoparasites that may transmit the infection. Primary prevention of trench fever relies on measures for avoiding infestation with body lice.
Bacillary angiomatosis is a proliferative vascular disease due to both B. quintana and B. henselae. The disease is characterised by angioproliferative lesions of skin (sarcoma-like skin lesions) or various organs (spleen, liver, bone marrow and lymph nodes). Bacillary angiomatosis due to B. quintana was mainly reported among immunodeficiency virus-infected persons in the 1990s, but can affect other immunocompromised individuals.
read more:
https://www.ecdc.europa.eu/en/bartonella-quintana-infection-trench-fever/facts
*
A laboratory animal surveillance program intended to screen for the presence of adventitious pathogens was performed at the Laboratory Animal Center of the Academy of Military Medical Sciences, Beijing, China.
It is unknown how the macaques were initially exposed to infected lice or how the lice became infected with B. quintana. We postulate that the lice became infected with B. quintana from an infected macaque and thereafter acted as efficient vectors among the rest of the macaques in the colony.
Four blood samples from 10 captive-bred rhesus macaques (Macaca mulatta) were presumed to be infected with Bartonella spp. according to Giemsa-stained smears and transmission electron microscopy . Further PCR and sequence analysis of 3 gene targets (internal transcribed spacer [ITS], gltA, and rnpB) confirmed the existence of B. quintana in the 4 parasite-positive macaques. In addition, B. quintana was successfully isolated from the 4 monkeys by blood plating.
read more …
https://wwwnc.cdc.gov/eid/article/19/2/12-0816_article
*
Bats are unique in their mobility as they are the only mammals capable of flight, allowing them to transmit EIDs during their foraging flights and during seasonal migrations. This extensive mobility, coupled with roosting plasticity and broad food range, means that bats could transport viral material to many different animal species in various locations per unit time. The ability to fly also has immunological implications. Flight requires a low body mass and bats have evolved to have hollow bones to decrease their body mass. The hollow bones allow them to fly, but as a result they do not have bone marrow as similar to non-volant mammals and must produce B-cells in different locations.
The bat origin has been confirmed (such as lyssaviruses, henipaviruses, coronaviruses), for other it has been suggested (filoviruses). Several recently identified viruses remain to be ‘orphan’ but have a potential for further emergence (such as Tioman, Menangle, and Pulau viruses). In the present review we summarize information on major bat-associated emerging infections and discuss specific characteristics of bats as carriers of pathogens (from evolutionary, ecological, and immunological positions). The majority of studies performed to date have focused on infections of significant public health and veterinary concern.
Severe acute respiratory syndrome (SARS) emerged during November 2002 in southern China, and a SARS coronavirus (SARS-CoV) was identified as the etiologic agent. This epidemic, and the identification of SARS-CoV in animals, associated with the wildlife trade in southern China particularly in civets and raccoon dog.
The level of nucleic acid sequence difference (∼8%) from SARS CoV in multiple genes was too great for SARS-like CoV in Rhinolophus bats to be the parent to the outbreak virus. The presence of multiple SARS CoV-like viruses, the inability to detect SARS CoV-like viruses in other species of wild animals, and the detection of a wide range of other coronaviruses in bats suggests that bats are a rich source of CoVs, however, the evolutionary pathway of SARS-CoV remains to be fully identified.
In China, high CoV prevalence was detected in the Vespertilionidae and Rhinolopidae families of bats. The overall prevalence was about 6%, but in certain bat colonies it was as high as 35–55%. Such diversity was significant and not only SARS-like CoVs (from Betacoronavirus, former group 2) but also additional putative novel subgroups from Alphacoronavirus (former group 1) were identified in bats. Similar results were reported from Hong Kong.
After the discovery of SARS-like CoVs in bats in Asia, a number of bat CoVs were identified in Europe, North America, South America, Australia, and Africa with an overall prevalence of 9 to 20%. In Europe, the alphacoronaviruses and betacoronaviruses, identified in Vespertilionidae bats, were genetically similar to the CoVs identified in bats from China . In the United States, 17% of Eptesicus fuscus and 50% of Myotis occultus were positive for CoVs. Phylogenetically these viruses belonged to the same alphacoronavirus group but formed distinct clusters from Asian CoVs. In Canada, an alphacoronavirus identified in Myotis lucifugus bat is probably a variant of alphacoronaviruses identified in Myotis occultus in the United States. In South America, the Trinidadain CoVs identified in Phyllostomidae bats were clustered with alphacoronavirus from North America. In Africa, enhanced surveillance demonstrated significant divergence of CoVs in bats from Kenya. In that study, SARS-like CoV was identified in a Chaerephon sp. bat (Molossidae). Furthermore, in contrast to China and Hong Kong, various CoVs were detected in bats from the families Hipposideridae and Pteropidae. Overall CoV prevalence in Kenya bats was approximately 19%, and CoV diversity was greater compared to that documented in Asia, Europe, North and South Americas, and Australia.
*
The primary drivers of bat-associated EIDs include overpopulation, environmental degradation, and socioeconomical forces. The emergence of new pathogens is associated with growth and increased density of humans and other mammals. In recent history, the human population has exploded, with an increase from 1 billion to 6.8 billion in the past 110 years. As populations grow, humans begin to inhabit previously untouched, often biodiverse areas. Research suggests that diseases are more likely to emerge in such regions. Coupled with this is the increased utilization of natural resources to meet the human demand for food and support consumption of goods.
Environmental degradation causes habitat disturbance and reduction, resulting in changes in species range and density. For example, land use changes such as mining and deforestation for farming and the construction of human habitats in the Amazon Basin have likely contributed to the re-emergence of vampire bat-derived rabies in humans. In 2004, at least 46 deaths were caused by vampire bat rabies (predominantly in Brazil and Colombia), whereas only 20 cases were transmitted by dogs in all of Latin America.
*
Human practices surrounding the production and consumption of food can contribute significantly to the risk of new pathogen emergence. For example, wet markets (in which live animals are sold and butchered on the spot) are an ideal environment for microbial exchange due to the high density of people, and the diversity of wild and farmed animals sold at these sites. Live animal markets appear to have contributed to the emergence of SARS in China in 2002. With primary bat origin, several intermediate hosts have been suggested (such as Chinese ferret-badgers, Melogale moschata, and raccoon-dogs, Nyctereutes procyonoides), although palm civets (Paguma larvata) are suspected to be the most important for the transmission of the virus to humans.
In Southern China, civets are both hunted and farmed for eating. In fact, civets tested in wet markets have a higher rate of seropositivity for SARS than those tested on farms, suggesting that the markets may serve as centers for viral transmission. In addition, food handlers and persons employed at wet markets are more likely to be seropositive than those with other occupations.
*
Rabies is an acute progressive encephalitis caused by viruses in the Genus Lyssavirus, Family Rhabdoviridae, with the highest fatality rate among conventional infectious diseases. Known in bats for well over a century, rabies is the best studied infection associated with the Chiroptera. Bats are the principal reservoirs for 10 of the 11 recognized lyssavirus species and are suspected as hosts of other putative species.
Another viral species, rabies virus (RABV), circulates in bats and other mammals (predominantly carnivores). Interestingly, RABV circulates in bats only in the Americas, whereas in carnivores, the disease circulates globally. In the Old World, bats maintain circulation of other lyssavirus species, such as Lagos bat virus (LBV), Duvenhage virus (DUVV), European bat lyssaviruses type 1 (EBLV-1) and 2 (EBLV-2), Australian bat lyssavirus (ABLV), Aravan virus (ARAV), Khujand virus (KHUV), Irkut virus (IRKV), West Caucasian bat virus (WCBV), and Shimoni bat virus (SHIBV). For these viruses, bats are the principal hosts, with only a few spillover infections documented in other mammals.
A paralytic disease in cattle and sporadically in humans bitten by a vampire bat has been reported from the time of the Spanish first colonized Latin America. However, the diagnosis of rabies was first confirmed by the identification of Negri bodies in the brain of cattle during an outbreak in Brazil in 1911. Vampire bats probably maintained rabies virus circulation for a long time prior to the arrival of Europeans in the Americas. The association between vampire bites and the disease was understood by natives, who cauterized or washed the bites to prevent the disease.
Economic losses due to vampire bat rabies in livestock are tremendous. In the enzootic area there is an at-risk population of more than 70 million head of cattle. Vampire bats usually bite many animals in a herd. The proportion of animals bitten may vary from 6 to 52% 15. Significant outbreaks of vampire bat rabies were documented in Amazon area (Brazil, Peru) during recent years. Up to 23–55% of respondents interviewed had vampire bat bites during the last year. During the outbreaks, up to 15% of such bites caused rabies in humans.
Another African bat lyssavirus, DUVV, is covered by rabies biologics, but still kills people because of insufficient knowledge, either in general public and health professionals. The DUVV is perhaps the most mysterious African lyssavirus. Of four isolates available, three came from humans who died of rabies after bat exposures and only one was isolated from an insectivorous bat, presumptive Miniopterus sp. The most recent human case occurred in 2007 in Kenya, where a Dutch tourist was attacked by a bat in a campsite of Tsavo West national park. The patient applied for medical help, but a local physician assured that bat rabies does not exist in Kenya and PEP was not administered. Several weeks later, back in the Netherlands, the patient developed rabies and died. The virus was identified as DUVV.
The discovery of ABLV in 1996 in the ‘rabies-free’ Australia was surprising. Following the discovery that flying foxes were a reservoir of Hendra virus, surveillance of these animals was increased. During this activity, ABLV was identified first in a sick black flying fox (Pteropus alecto). The second case was diagnosed retrospectively in another bat of the same species, sampled in 1995 with signs of unusual aggressiveness. Later ABLV was documented in each of the four flying fox species, present in continental Australia.
https://www.tandfonline.com/doi/full/10.3402/ehtj.v4i0.7159
*
To predict the next CoV that will cause a virus outbreak in future, we list the general factors that may contribute to this outbreak. Firstly, bats host a large number of highly diverse CoVs. It is known that CoV genomes regularly undergo recombination during infection, and a rich gene pool can facilitate this process. Secondly, bat species are widely distributed and live close to humans. Thirdly, the viruses are pathogenic and transmissible. In this context, SADS-CoV and SARS-CoV outbreaks in China are not unexpected. By this model, there are other CoVs that have not yet caused virus outbreaks but should be monitored.*
As a coronavirus spreads in China and around the world, scientists are scrambling to find out exactly where it came from. Now, a new study provides more clues to the virus’ origins, and points to bats as the most likely hosts. To learn more about the virus’ origins, the researchers compared the 2019-nCoV genetic sequence with those in a library of viral sequences, and found that the most closely related viruses were two coronaviruses that originated in bats; both of those coronaviruses shared 88% of their genetic sequence with that of 2019-nCoV. (When compared with two other coronaviruses known to infect people — SARS and MERS — 2019-nCoV shared about 79% of its genetic sequence with SARS and 50% with MERS.)
*
A-year-old report had warned China of a potential coronavirus outbreak. In the report, Chinese scientists called for urgent countermeasures to stop such an outbreak. Our Eum Ji-young has more. It turns out a Chinese research team warned the medical world last year of the potential for the outbreak of coronaviruses from bats.
Abstract
During the past two decades, three zoonotic coronaviruses have been identified as the cause of large-scale disease outbreaks–Severe Acute Respiratory Syndrome (SARS), Middle East Respiratory Syndrome (MERS), and Swine Acute Diarrhea Syndrome (SADS). SARS and MERS emerged in 2003 and 2012, respectively, and caused a worldwide pandemic that claimed thousands of human lives, while SADS struck the swine industry in 2017.
*
They have common characteristics, such as they are all highly pathogenic to humans or livestock, their agents originated from bats, and two of them originated in China. Thus, it is highly likely that future SARS- or MERS-like coronavirus outbreaks will originate from bats, and there is an increased probability that this will occur in China. Therefore, the investigation of bat coronaviruses becomes an urgent issue for the detection of early warning signs, which in turn minimizes the impact of such future outbreaks in China.
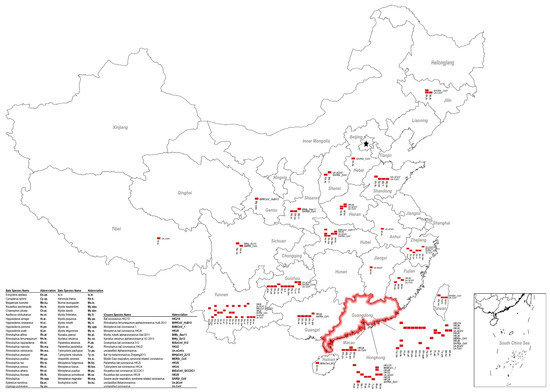
The purpose of the review is to summarize the current knowledge on viral diversity, reservoir hosts, and the geographical distributions of bat coronaviruses in China, and eventually we aim to predict virus hotspots and their cross-species transmission potential.
Fifteen years after the first highly pathogenic human coronavirus caused the severe acute respiratory syndrome coronavirus (SARS-CoV) outbreak, another severe acute diarrhea syndrome coronavirus (SADS-CoV) devastated livestock production by causing fatal diseases in pigs.
Both outbreaks began in China and were caused by coronaviruses of bat origin . This increased the urgency to study bat coronaviruses in China to understand their potential of causing another virus outbreak.
In this review, we collected information from past epidemiology studies on bat coronaviruses in China, including the virus species identified, their host species, and their geographical distributions. We also discuss the future prospects of bat coronaviruses cross-species transmission and spread in China.
>
SADS-CoV was recently identified as the etiological agent responsible for a large-scale outbreak of fatal disease in pigs in China that caused the death of more than 20,000 piglets.
Bats were linked to a few highly pathogenic human diseases, supporting this hypothesis. Some of these well characterized bat viruses, including bat lyssaviruses (Rabies virus), henipaviruses (Nipah virus and Hendra virus), CoVs (SARS-CoV, MERS-CoV, and SADS-CoV), and filoviruses (Marburg virus, Ebola virus, and Mengla virus), pose a great threat to human health.
A comprehensive analysis of mammalian host–virus relationships demonstrated that bats harbor a significantly higher proportion of zoonotic viruses than other mammalian orders. Viruses from most of the viral families can be found in bats.
The most recent SADS-CoV spillover was traced back to bats [2]. In addition, bats also carry α-CoVs that are related to pathogenic human 229E- and NL63-CoVs, as well as pandemic swine coronavirus PEDV . Bats carry major α- (10 out of 17) and β- (7 out of 12) CoV species that may spillover to humans and cause disease . Attributed to the wide distribution of bats, CoVs can be found worldwide, including China.
Two bat CoVs caused outbreaks in China; it is thus urgent to study the reasons to avoid future outbreaks. China is the third largest territory and is also the most populous nation in the world. A vast homeland plus diverse climates bring about great biodiversity including that of bats and bat-borne viruses—most of the ICTV coronavirus species (22/38) were named by Chinese scientists studying local bats or other mammals. The majority of the CoVs can be found in China
In November 2012, the first case of SARS was recorded in Foshan city, Guangdong Province, China. In 2005, two independent Chinese groups reported the first bat SARS-related CoV (SARSr-CoV) that was closely related to human SARS-CoV, implying a bat origin of the latter.
Since then, more bat SARSr-CoV isolates were identified in China. Genome identities of these bat SARSr-CoVs are as high as 92% to human SARS-CoV, but their major receptor binding spike proteins cannot use the human virus entry receptor ACE2. Whether they are the progenitor viruses of SARS-CoV is debatable.
In 2013, the isolation of a bat SARSr-CoV that uses the ACE2 receptor provided the strongest evidence of the bat origin of SARS-CoV . Furthermore, the building blocks for SARS-CoV were identified from eleven different SARSr-CoV viral strains in a five-year surveillance program in a cave inhabited by multiple species of horseshoe bats in Yunnan Province, China.
https://www.mdpi.com/1999-4915/11/3/210/htm
*
This paper predicted coronavirus outbreak one year before it happened